Mahidol University Joins Press Conference on Achievements of Research and Innovation on "Cold Chain Logistics"
On May 27, 2021 Clinical Professor Emeritus Piyasakol Sakolsatayadorn, M.D., Chairman of the Mahidol University Council, and Assoc. Prof. Dr. Duangpun Singkarin, Faculty of Engineering, Mahidol University jointly announced the achievements of the research and innovation on “Cold Chain Logistics: Cold Chain Development of COVID-19 Vaccine for Temperature Control and Traceability in Transportation and Storage”.
The event was led by Special Professor Dr. Anek Laothamatas, Minister of Higher Education, Science, Research and Innovation (MHESI), Professor Emeritus Dr. Soottiporn Chittmittrapap, Chairman of Thailand Science, Research and Innovation Promotion Committee, Dr. Kornkrit Limsommut, Director of Department of Public Health Administration, and Dr. Wiparat De-Ong, Director of the National Research Office.
The National Research Council of Thailand (NRCT), Ministry of Higher Education, Science, Research, and Innovation organized this event to disseminate research results and innovations that are ready to be used at the Ministry of Higher Education, Science, Research and Innovation and through Zoom online video conferencing.
Clinical Professor Emeritus Piyasakol Sakolsatayadorn, M.D., Chairman of the Mahidol University Council, said, “As the novel coronavirus 2019 (COVID-19) outbreak has spread in Thailand and many countries around the world, the most effective way to end the epidemic is to produce a vaccine that is resistant to the COVID-19 virus and to manage the distribution of vaccines for efficient supply according to the requirements of Cold Logistics Supply Chain Management System. Therefore, it is a key principle to help develop the transport design and vaccine cold chain traceability system that can control temperature throughout the transportation period. The idea was to develop a COVID-19 vaccine traceability system so that it can be used in real-world situations of vaccine transportation. Also, it was to ensure that the vaccine is stored and transported effectively; it is safe for vaccine users with the tracking system "Cold Chain" of COVID-19 Vaccine and the vaccine distribution system”.
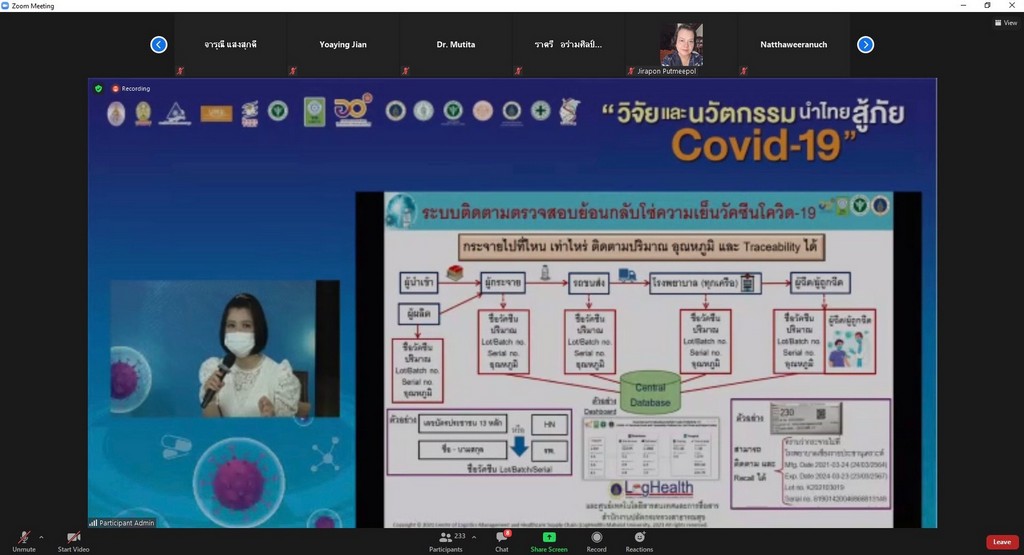
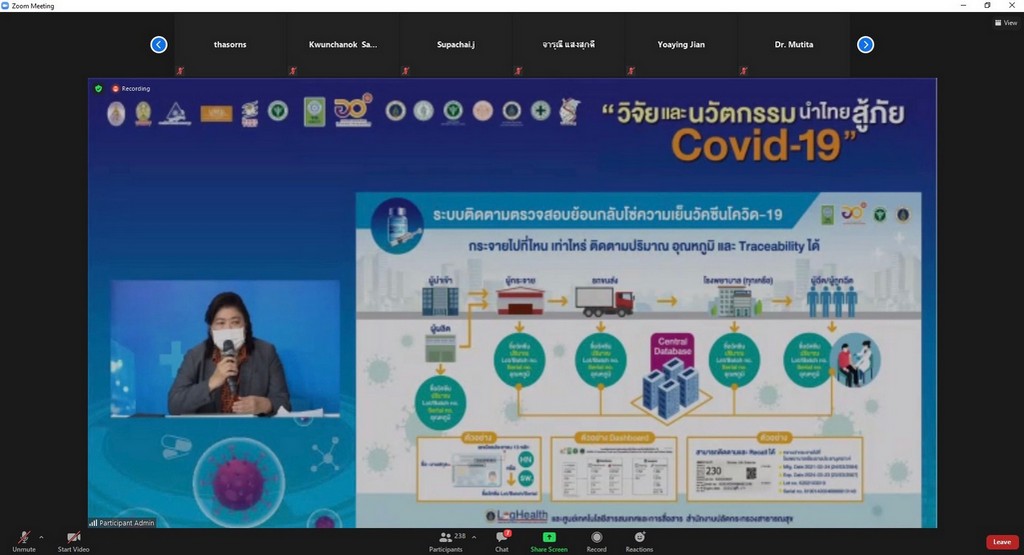
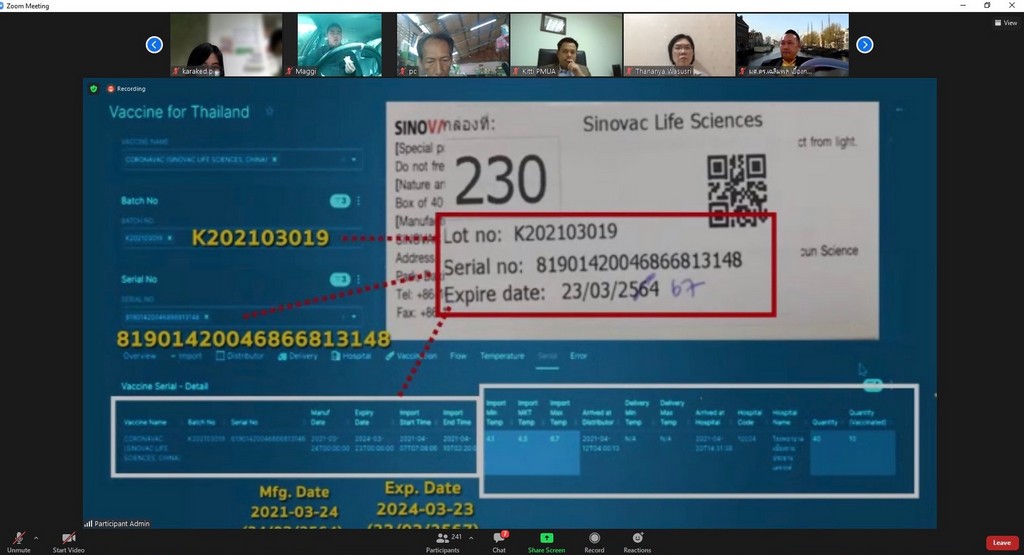
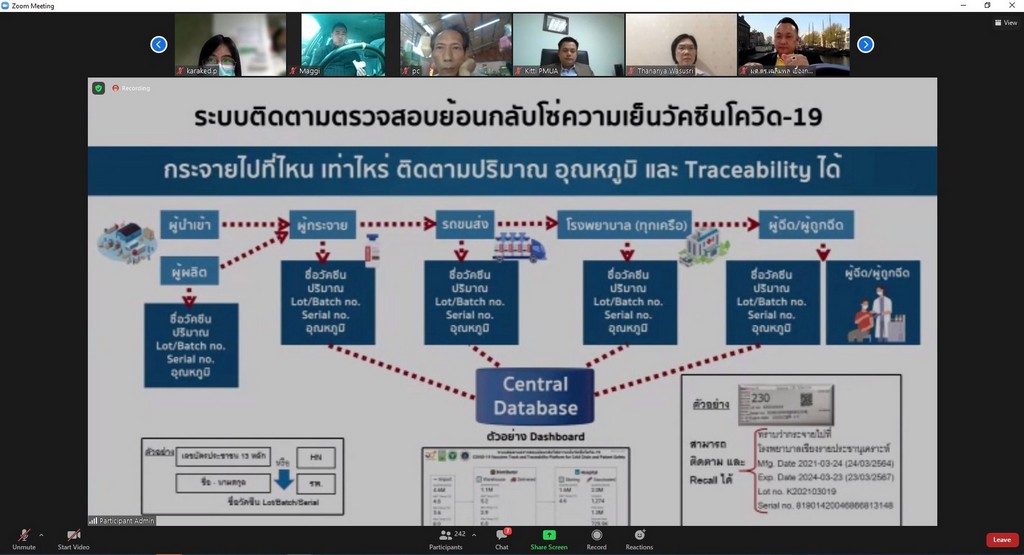
Assoc. Prof. Dr. Duangpun Singkarin, Faculty of Engineering, Mahidol University and the Research Project Leader stated that, “This platform connects and collects information from all stakeholders. All vaccine details will be displayed regarding the time of the production of vaccines domestically and importing the vaccine from other countries, the storage of the vaccine, transport and distribution of the vaccine with whom, when, and where. The data includes cold control information and the temperature levels throughout the supply chain, from the manufacturer or carrier to the hospital service or provider and to the public as recipients”.
“This is for the benefit of people and personnel and to ensure the quality of the vaccine and its efficiency. The data set of "Cold Chain" COVID-19 Vaccine will be entered in the Ministry of Public Health's Central Co-Vaccine System. This information includes the registration of the manufacturer or importer of the vaccine as well as vaccine service recipients. We will use the data from the Information and Communication Technology Center Office of the Permanent Secretary, Ministry of Public Health”, Assoc. Prof. Dr. Duangpun Singkarin mentioned.
“The doctor application is ready to show the results of Supply Chain and Logistics. The "Cold Chain" COVID-19 Vaccine can be traced and checked. If there is a problem, e.g., the illegibility of vaccine expiry date, it will be diagnosed and corrected. The COVID-19 Vaccines Track and Traceability Platform will be able to find out where the vaccine is and who is injected. So, the vaccine can be provided in time if there is a problem regarding vaccine storage. By using a cold chain system, it is necessary to maintain a constant temperature. The vaccines currently imported in Thailand are AstraZeneca (AstraZeneca) and Sinovac. A constant temperature of 2-8 degrees must be maintained throughout the supply chain with a sensor monitoring system to monitor the cooling level and store the data. A vaccine cooler must be available in the hospital.”
“Moreover, the COVID-19 vaccine cold chain monitoring-traceability system is able to link data throughout the cold chain via IoT system regarding the product information and vaccine doses produced, imports or purchases, storage temperature, number and type of vaccines distributed to each hospital, and vaccine recipient information. Each box of the vaccine is identified by a serial number to prevent mistakes. We try to build confidence in people and go through the crisis at the same time”, she continued.
Many organizations have collaborated in this project, including the National Research Agency (NCC) with supporting the budget for developing this system, Subcommittee for Administration of Coronavirus Vaccination 2019, Ministry of Public Health, Food and Drug Administration (FDA), Government Pharmaceutical Organization (GPO), Department of Medical Sciences, Department of Disease Control , Importers, Vaccine Carriers, Health Administration Division Office of the Permanent Secretary and Information and Communication Technology Center Office of the Permanent Secretary, Ministry of Public Health, Hospitals under the Ministry of Public Health, and the Private Sector, including the Federation of Thai Industries, AI and Robotics Ventures (ARV), and Nova Green Power System Co., Ltd.
Click to watch the video on Cold Chain Logistics:
https://www.youtube.com/watch?v=MRDhW9xxJ18
Click to login to Cold Chain Logistics platform:
https://datastudio.google.com/u/0/reporting/731713b6-a3c4-4766-ab9d-a6502a4e7dd6/page/JMn3B